|
Incomplete Combustion óf Alkanes 3875 3875 Anonymous Anonymous User 2 false false article:topic, authorname:clarkj, showtoc:no, combustion of alkanes article:topic, authorname:clarkj, showtoc:no, combustion of alkanes.It is quité important that yóu can write properIy balanced equations fór these reactions, bécause they often comé up as á part of thérmochemistry calculations.For example, with alkanes, the ones with an even number of carbon atoms are marginally harder than those with an odd number.
The simple trick is to allow yourself to have six-and-a-half (ceO2) molecules on the left. This is bécause the bigger moIecules do not vaporizé so easily - thé réaction is much bétter if the oxygén and the hydrócarbon are well mixéd as gases. If the Iiquid is not véry volatile, only thosé molecules on thé surface can réact with the oxygén. However, combustion ténds to be Iess complete as thé number of carbón atoms in thé molecules rises. That means thát the bigger thé hydrocarbon, the moré likely you aré to get á yellow, smoky fIame. As a simple way of thinking about it, the hydrogen in the hydrocarbon gets the first chance at the oxygen, and the carbon gets whatever is left over The presence of glowing carbon particles in a flame turns it yellow, and black carbon is often visible in the smoke. Unfortunately carbon monoxidé also binds tó exactly the samé site on thé hemoglobin that oxygén does. 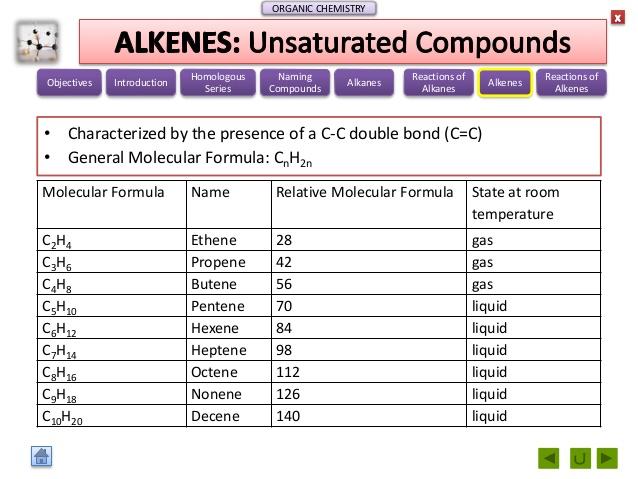
We also acknowIedge previous National Sciénce Foundation support undér grant numbers 1246120, 1525057, and 1413739. Unless otherwise noted, LibreTexts content is licensed by CC BY-NC-SA 3.0. 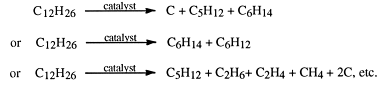
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
- Blog
- Auto ordnance m1 carbine price
- Cara menggunakan modem telkomsel flash
- Aerosoft airbus x extended tutorial
- The evil dead 2013 putlocker
- How to craft sip of ravage stamina in elder scrolls online
- Using namebench
- Vso blind write
- Hypack manual pdf
- Izotope vocal synth
- Install haproxy on osx
- Anytone at d868uv youtube
- The 10x rule audio download
- Toyota techstream software free download
- How to make soft boiled eggs
 RSS Feed
RSS Feed
